
Who is eligible to get the Novavax COVID-19 vaccine? These conditions are typically treatable, so it’s important to seek medical attention right away if symptoms develop. Symptoms typically start within 10 days of a dose and include chest pain, shortness of breath, and fast or pounding heartbeat. Myocarditis and pericarditis: Although rare, myocarditis and pericarditis have been reported with the Novavax COVID-19 vaccine. Call 911 or seek immediate medical attention if you experience difficulty breathing, swelling of the face, tongue, or throat, and/or a bad rash on your body. Allergic reactions usually happen within this timeframe, but they can also happen a few hours later. These include severe allergic reactions (including anaphylaxis), myocarditis (heart inflammation), and pericarditis (heart lining inflammation).Īllergic reactions: After your shot, you may be asked to wait around for about 15 to 30 minutes to be monitored for a reaction. While rare, serious side effects have been reported with the Novavax COVID-19 vaccine. Are there any serious side effects to know about? Let them know if any side effects don’t go away or start getting worse. This may include over-the-counter products, like acetaminophen (Tylenol) or ibuprofen (Advil, Motrin). If any side effects are bothersome, talk to your healthcare provider about medications you can take for relief. Injection-site reactions (pain, redness, swelling) 
Similar to the other COVID-19 vaccines, the Novavax COVID-19 vaccine can cause mild side effects that typically go away after a few days. 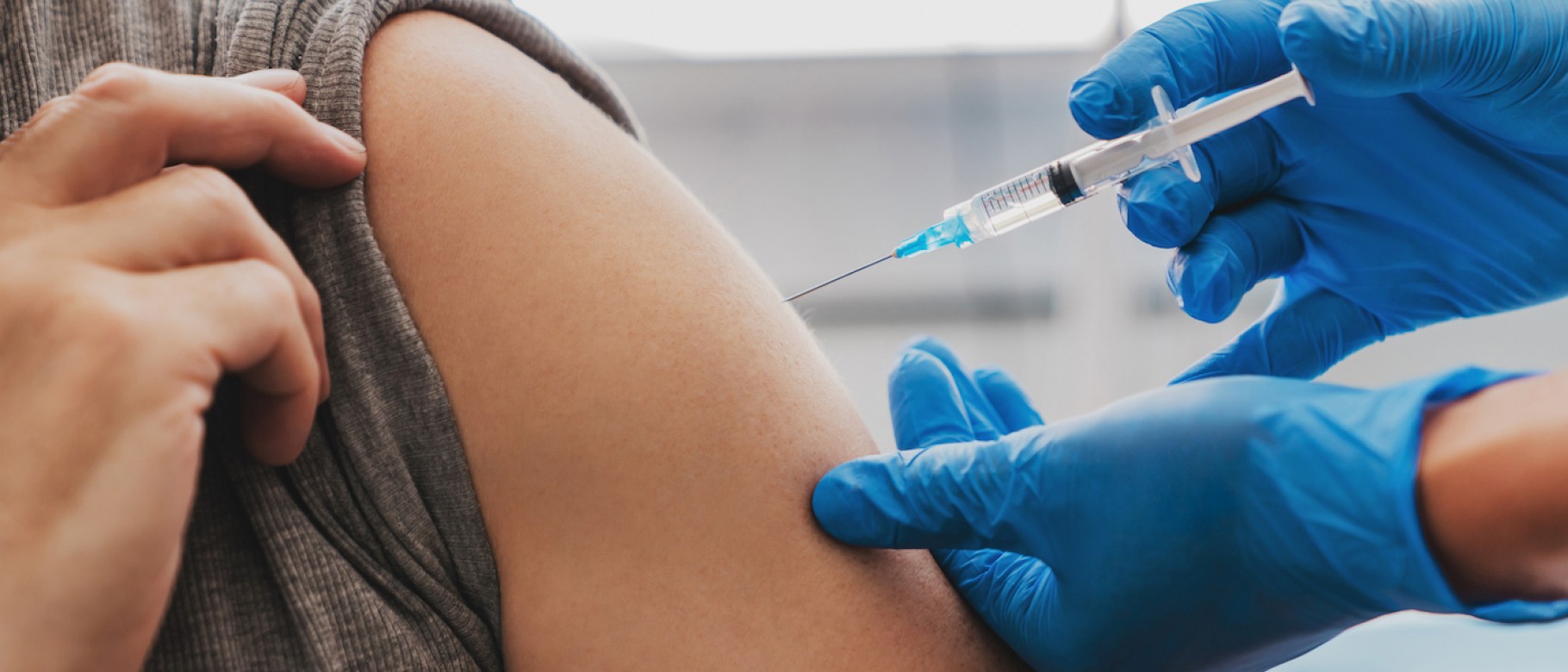
What are the known side effects of the Novavax COVID-19 vaccine? As of January 2023, the Omicron XBB.1.5 and BQ.1.1 subvariants make up most U.S. We don’t have much information about how well the Novavax COVID-19 vaccine works against newer, prevalent subvariants. Even so, the vaccines have been proven to be a valuable tool in preventing serious illness and death from COVID-19. However, as SARS-CoV-2 continues to produce new variants, COVID-19 vaccine effectiveness seems to be a moving target. It was also 100% effective against moderate to severe disease. During this time, the vaccine was about 90% effective against symptomatic COVID-19. The Novavax vaccine clinical trials initially captured information from late January through April 2021. How effective is the Novavax COVID-19 vaccine? 
It doesn’t matter which COVID-19 vaccine you received for your primary series. After receiving your first dose, you’ll wait 3 weeks before receiving your second dose. Currently, the same version of the vaccine is used for the primary series and booster shot.Īdults who’ve completed their primary vaccine series at least 6 months ago are eligible to receive a Novavax booster shot. And it’s given as an injection into the muscle of your upper arm. Like the Pfizer and Moderna vaccines, the Novavax COVID-19 vaccine is given as a two-dose primary series. It’s also authorized for a single booster shot.Įach Novavax shot contains 5 mcg of spike protein. How is the Novavax COVID-19 vaccine dosed? This is more conventional vaccine technology compared to the Pfizer, Moderna, and J&J COVID-19 vaccines. In fact, vaccines like the hepatitis B, HPV, and shingles vaccines all fall into this category. Protein-based vaccines have been around for a while. It attacks the protein, creating antibodies that can recognize and fight the real virus if you’re infected in the future. Adjuvants are added to a variety of vaccines to help them work better.Īfter receiving a vaccine dose, your immune system sees the foreign protein and knows it doesn’t belong. So, it’s combined with an adjuvant - a substance that helps boost your immune response against it. Your immune system may not respond as strongly to the spike protein on its own. In this case, it’s the spike protein of SARS-CoV-2, the virus that causes COVID-19. This type of vaccine contains an antigen (protein) that’s unique to a germ. The Novavax COVID-19 vaccine is an adjuvanted protein-based vaccine. How does the Novavax COVID-19 vaccine work? Here, we’ll cover some of the basics you should know. Since then, the Novavax COVID-19 vaccine has been authorized for additional age groups and boosters. In these cases and more, the Novavax COVID-19 vaccine provides another option to consider. And others have severe allergies that conflict with current COVID-19 vaccine options. Because of this, some people have been hesitant to get vaccinated. The Pfizer, Moderna, and Johnson & Johnson (J&J) COVID-19 vaccines use newer vaccine technology. So, you may be wondering why a fourth vaccine option was authorized for this age group. adults had received at least one COVID-19 vaccine dose. In July 2022, the FDA authorized the Novavax COVID-19 vaccine for adults.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
